White Papers
Heavy Metal Detection in Water

Water pollution is a serious worldwide issue affecting various aspects of society, such as human/animal healthcare, the food chain, and the tourism industry; therefore, the improvement of water quality can encourage the economic growth of a nation and contribute significantly to inequality reduction. Accessing clean and safe water for daily consumption, such as drinking, cooking, personal hygiene, and sanitation, is a fundamental human right. However, according to the World Health Organization (WHO) report, there are 772 million people who lack even essential drinking water services and 122 million people who depend on surface water. Globally, at least 2 billion people use contaminated drinking water sources [1]. Contaminated water can cause numerous health concerns, including diarrhea, cholera, dysentery, typhoid, and polio [2]. Heavy metals are one of the primary sources of water pollution. They are persistent in the environment once discharged, which is a significant cause of concern for the toxicity of the water. Therefore, heavy metals must be removed from the water source in the water treatment process to ensure clean drinking water.
Heavy metals are metallic chemical elements with relatively high density (>5 g cm-3) [3]. Most heavy metals or specific forms of these elements are even classified as human carcinogens that threaten both ecological balance and human health at trace levels, such as arsenic (As), cadmium (Cd), chromium (Cr), lead (Pb), and mercury (Hg) [4]. The ongoing expansion of heavy metal pollution results from several factors, including natural processes or pollutants discharged into the environment—for example, in natural processes cases, As is released from weathered rock and contaminates groundwater aquifers [5]. Meanwhile, the development of industrial activities is a primary source to inevitably contributes to heavy metal contamination in the environment. Especially, hazardous substances and controlled chemicals used in the production process consisting of Pb, Cd, Hg, and Cr have been reported [6]. In general, Pb, Cd, Hg, and Cr could be presented in water as a result of emission from the plant, Pb from a battery plant , Cr from an electroplating plant, and Hg and Cd from a power plant.
Moreover, the toxicity of heavy metals usually depends on the oxidation states of each element. For example, the inorganic compound of As(III) is generally considered to be more toxic than As(V) up to 60-fold. Besides, Cr(III) is regarded as an essential element for mammals that plays a major role in insulin, lipid, and glucose metabolism.
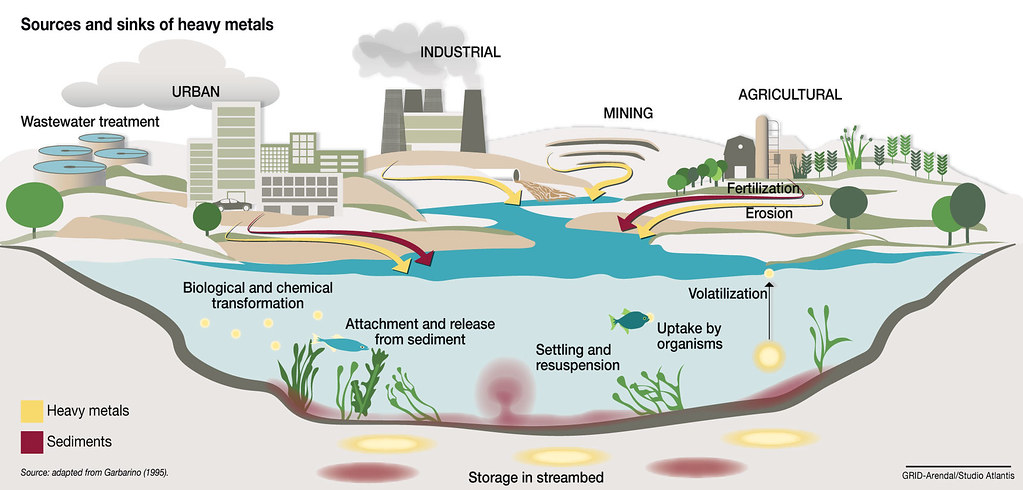
Figure 1 Sources and sink of heavy metals
Therefore, various industrial processes can produce problematic heavy metal concentrations in discharged water from factories that are harmful to humans and can contaminate agricultural land. The source and sink diagram of heavy metal are presented in Figure 1. Testing for heavy metals at the part per billion (ppb) level is essential to meet established international limits. The maximum permissible concentration of heavy metals in wastewater has been regulated by various regulatory authorities.
The conventional techniques for heavy metal detection comprise atomic absorption spectroscopy (AAS) [8, 9], flame or furnace spectroscopy [10], inductively coupled plasma optical emission spectroscopy (ICP−OES) [11, 12], inductively coupled plasma mass spectrometry (ICP−MS) [13, 14], and energy dispersive X-ray fluorescence (EDXRF) spectrometry [15]. Although these techniques are highly sensitive and selective to the trace level of heavy metals, there are some limitations, including being operated by a trained scientist and needing more field test portability due to their bulky and expensive instruments.
Extensive sample preparation in high sample volume might be required and need a long time to run. A single mislabeled sample from outside laboratory service can result in extensive downtime. Moreover, their operating costs are also high. Since heavy metal pollution in the environment is constantly increasing due to the growth of industrial development and their high toxicity that threatens the ecological balance and human health, there is a high demand for the development of a rapid, portable, and on-spot automated detection system. Compared with traditional methods, electrochemical detection of heavy metal ions is a very promising strategy that has attracted widespread attention due to its portability, fast analysis, and low cost. Therefore, the electrochemical instrument can be adapted to various on-site inspection sites and applied in agriculture, life science, clinical diagnosis, and analysis.
Electrochemical Detection
Electroanalytical methods have been extensively applied in sensing and biosensing. An electroanalytical sensor is based on electrochemical transduction that employs a two- or three-electrode arrangement, as shown in Figure 2. Electrochemical transductions need the potentiostat, which is an electronic instrument to control the potential between its electrode and measure the current flow through the chemical cell. The principle of the electrochemical method is to study the chemical process that results in electron transfer. The electron exchange phenomenon produces a form of electrical change, i.e., current, potential, charge, and so on, which could be generated by such a process between the electrode and electrolyte known as an oxidation-reduction (redox) reaction. For the electroanalysis of heavy metal, anodic stripping voltammetry is widely used to quantify the concentration of heavy metal. In the electrodeposition step, the constant potential is applied over a period of time for the accumulation of the target analyte onto the electrode surface by a reduction reaction (M+ + e- → M(0)) before the stripping step. According to the heavy metal preconcentration process, the response signal of heavy metal exhibits a high stripping current using the anodic or cathodic stripping method. In the stripping method, electrochemical techniques such as linear scan voltammetry, differential pulse voltammetry, square wave voltammetry, etc., could be performed to measure the concentration of heavy metal. The experimental result is presented in a linear relationship between the stripping current vs. the concentration of heavy metal.



